Most of the locally defined terms in this glossary are ones that have significant and particular application to human life expansion. Many of them have one or more technical meanings within the sciences related to life expansion that are different from that of the common usage, or even within other sciences. In such cases, we have chosen to include only the meaning(s) relevant to the subject(s) covered within MoreLife. In constructing our definitions we wish to acknowledge the value of these references.
In order to prevent unnecessary duplication, we have selected several excellent sources for external definitions. However, since our usage of some terms is quite specialized and we wish to emphasize certain parts of their meanings, we often locally define terms from these dictionaries within this glossary. In addition, because many anatomical parts and various specific diseases may be of interest to the reader, links to well presented glossary information on other websites have been provided.
| GABAergic |
1) describing a nerve or other cell that is acted upon by 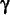 -aminobutyrate (or -aminobutyrate (or 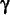 -amino-n-butyric acid - GABA. -amino-n-butyric acid - GABA.
2) Describing nerves that act by releasing 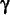 -aminobutyrate. -aminobutyrate. |
| galacto- |
prefix (in chemical nomenclature) indicating a particular configuration of a set of (usually) four contiguous CHOH groups, as in the acyclic form of D- or L-galactose. |
galactose
brain sugar
cerebrose
galactolipid |
symbol: Gal; the trivial name for the aldohexose, galacto-hexose; there are two enantiomers, D- and L-galactose. D-galactose Isymbol: D-Gal), commonly known as galactose, and formerly known as brain sugar or cerebrose, rarely occurs free but is widely distributed in combined form in plants, animals, and microorganisms as a constituent of many oligosaccharides and polysaccharides; it occurs also in galactolipids (any glycolipid containing one or more residues of galactose and/or N-acetylgalactosamine) and as its glucoside in lactose. |
gamma globulin
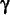 -globulin -globulin |
Any of a band of serum proteins that migrate most slowly on electrophoresis. In humans this normally constitutes quantitatively the largest fraction of the total globulins. Since the 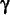 -globulins are predominantly anti-bodies, and are not synthesized in the liver as are virtually all the other serum proteins (of which albumin is the largest fraction), the albumin/globulin ratio has traditionally been a useful indicator of liver function. The -globulins are predominantly anti-bodies, and are not synthesized in the liver as are virtually all the other serum proteins (of which albumin is the largest fraction), the albumin/globulin ratio has traditionally been a useful indicator of liver function. The
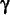 -globulins may be separated from the other serum globulins in pooled adult serum and may then be used for administration by intramuscular injection to provide passive immunity in premature infants or for those with cases of infectious hepatitis. -globulins may be separated from the other serum globulins in pooled adult serum and may then be used for administration by intramuscular injection to provide passive immunity in premature infants or for those with cases of infectious hepatitis. |
gamma glutamyl cycle
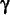 -glutamyl cycle -glutamyl cycle |
An integrated cycle of chemical events that uses the 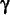 -carboxyl group of glutamate to transport amino acids actively across (mammalian) cell membranes. Glutathione supplies the activated -carboxyl group of glutamate to transport amino acids actively across (mammalian) cell membranes. Glutathione supplies the activated 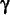 -glutamyl group, which reacts by transpeptidation with the amino acid to be transported. The resulting -glutamyl group, which reacts by transpeptidation with the amino acid to be transported. The resulting 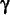 -glutamylamino acid enters the cell, where it is split into the free amino acid and 5-oxoproline. The latter is then converted to glutamate in an ATP-dependent reaction. The cysteinylglycine formed from glutathione in the initial transpeptidation step is hydrolyzed by a peptidase. Glutathione is then regenerated in two ATP-dependent steps. -glutamylamino acid enters the cell, where it is split into the free amino acid and 5-oxoproline. The latter is then converted to glutamate in an ATP-dependent reaction. The cysteinylglycine formed from glutathione in the initial transpeptidation step is hydrolyzed by a peptidase. Glutathione is then regenerated in two ATP-dependent steps. |
gamma radiation
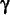 -radiation -radiation
transformation (nuclear) |
Electromagnetic radiation with wavelengths in the range 0.1-100 pm (picometers -
10-12) and frequencies 3-3000 EHz (1018) emitted by some atomic nuclei during transformation (change of one nuclide into another). Gamma rays have shorter wavelengths and higher photon energies than X-rays and can, thus, be more damaging to cells. |
+gen
+ogen
precursor
+genesis |
A combining form, or (after a consonant) +ogen, signifying producing or capable of producing (either directly or indirectly). In biological sciences it is used especially
(1) with word-stems relating to physiological or pathological processes, states, conditions, etc., to denote a causative agent (eg. antigen, mutagen, estrogen);
(2) with names of a certain subclass of protease or blood components, to denote their inactive precursors (substances from which they can be derived by enzymic action) eg. angiotensinogen, caseinogen, fibrinogen, trypsinogen);
(3) with names or name-roots of certain metabolites, to denote either a storage substance (eg. glycogen, phosphagen) or a biosynthetic precursor (eg. porphobilinogen (of polyphyrins);
(4) with word-stems relating to physical properties (eg. chromogen).
+genesis, combining form denoting beginning or origin; development; generation. |
| glucagon |
A 3.5 kDa. single-chain, 29 residue hormone synthesized in the A cells of pancreatic islets and mammalian (except human) gastric mucosa. It is secreted in response to falling blood glucose levels and has an opposing action to that of insulin by increasing the glucose concentration in the blood. Its effects include the stimulation of glycogenolysis and gluconeogenesis in liver, increased lipolysis in adipose tissue, and increased insulin secretion. |
gluco+
gluco-
|
Or (sometimes before a vowel) gluc+ combining form indicating glucose. gluco- prefix (in chemical nomenclature) indicating a particular configuration of a set of (usually) four contiguous CHOH groups as in the acyclic forms of D- or L-glucose. See monosaccharide |
glucogenesis
glucogenic |
The production of glucose in an organism, especially from noncarbohydrate precursors.
Glucogenic, describing a substance, some or all the carbon atoms of which can be used to produce glucose in an organism. In particular, a glucogenic amino acid is any amino acid that gives rise to increased urinary glucose excretion when fed to a diabetic animal. |
gluconeogenesis
gluconeogenic pathway |
The metabolic formation of carbohydrates from non-carbohydrate organic precursors in the liver. Formerly the term was restricted to the metabolic formation of glucose from noncarbohydtate precursors. Physiologically, an important source of precursor molecules is amino acid released from muscle protein during fasting. This is under hormonal control especially by glucocorticoids. The amino acid, alanine is an important precursor, and there is extensive conversion in muscle of other amino acids to alanine, which is then transported to the liver, where it undergoes transamination to form pyruvate. Pyruvate is then successively converted to various intermediates in the gluconeogenic pathway to glucose or other sugars. This pathway in the liver is responsible for the conversion of non-carbohydrate precursors to glucose 6-phosphate and thence to glucose or other carbohydrates. |
glucose
grape sugar
corn sugar
dextrose |
symbol: Glc; the trivial name for the aldohexose, gluco-hexose; there are two enantiomers, D- and L-glucose. D-(+)-Glucose (symbol: D-Glu), commonly known as glucose, and formerly known as grape sugar or corn sugar, is dextrorotatory, hence also formerly (and sometimes still) known as dextrose; it is 0.74 times as sweet as sucrose. Aged aqueous solutions of D-glucose contain an equilibrium mixture of 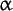 -D-glucopyranose, -D-glucopyranose, 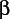 -D-glucopyranose, and the open-chain form (see also mutarotation), whereas in crystals -D-glucopyranose, and the open-chain form (see also mutarotation), whereas in crystals 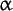 -D-glucopyranose monohydrate is the stable form below 50°C. Combined D-glucose may have either the -D-glucopyranose monohydrate is the stable form below 50°C. Combined D-glucose may have either the 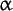 or or 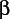 configurations, but is invariably in the pyranose form. D-Glucose is an important source of energy for living organisms. It is found free in fruits and other parts of plants, in honey, and in animals, especially in the blood (~5 mM in human blood). In combined form it occurs in many homo- and hetero-oligosaccharide and polysaccharides, especially in the animal storage polysaccharide, glycogen, and in the plant storage polysaccharides, cellulose and starch. configurations, but is invariably in the pyranose form. D-Glucose is an important source of energy for living organisms. It is found free in fruits and other parts of plants, in honey, and in animals, especially in the blood (~5 mM in human blood). In combined form it occurs in many homo- and hetero-oligosaccharide and polysaccharides, especially in the animal storage polysaccharide, glycogen, and in the plant storage polysaccharides, cellulose and starch. |
| glucose tolerance test |
Any procedure designed to assess the response of an individual to a loading dose of glucose, widely used in the diagnosis of diabetes mellitus. The standard oral glucose tolerance test is performed in the morning following at least 3 days' unrestricted diet, physical activity, and freedom from medication. The subject should have fasted for 12 hours before the test and remain seated and non-smoking during the test. After collection of the fasting blood sample, 75g (or 1.75g/kg) of body weight for children) glucose is ingested in 5 minutes in 200-500 mL water (formerly loads of 50g and 100g glucose were used). Blood samples are then collected at 30 minute intervals for 2 hours, and urine samples are collected at 0, 1, and 2 hours. All samples are assayed for glucose. For capillary blood samples, a maximum glucose level below 10 mmol/L (180mg/100mL) and 2-hour value less than 7.5 mmol/L (135mg/100mL) are the criteria for normality. If venous blood is used, the corresponding values are 8.9 mmol/L (160 mg/100mL) and 6.1 mmol/L (110 mg/100mL). The urine samples should be glucose free.
An intravenous glucose tolerance may be used when abnormal carbohydrate absorption from the intestines is suspected. |
| glutathione |
Abbr.: GSH; symbol: Glu(-Cys-Gly); 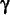 -glutamyl-cysteinylglycine, a tripeptide that is widely distributed in most if not all cells. It acts as a coenzyme for some enzymes and as an antioxidant in the protection of sulfhydryl groups in enzymes and other proteins; it has a specific role in the reduction of hydrogen peroxide and oxidized ascorbate, and it participates in the -glutamyl-cysteinylglycine, a tripeptide that is widely distributed in most if not all cells. It acts as a coenzyme for some enzymes and as an antioxidant in the protection of sulfhydryl groups in enzymes and other proteins; it has a specific role in the reduction of hydrogen peroxide and oxidized ascorbate, and it participates in the 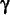 -glutamyl cycle. Glutathione may be oxidized with the formation of a disulfide bridge linking two molecules and generation of the compound known as oxidized glutathione (or the oxidized form of glutathione)(abbr.: GSSG). For clarity, glutathione is sometimes termed reduced glutathione (or the reduced form of glutathione). -glutamyl cycle. Glutathione may be oxidized with the formation of a disulfide bridge linking two molecules and generation of the compound known as oxidized glutathione (or the oxidized form of glutathione)(abbr.: GSSG). For clarity, glutathione is sometimes termed reduced glutathione (or the reduced form of glutathione). |
glycan
polysaccharide
homoglycan
heteroglycan
glucan
glycosan |
Alt. name, polysaccharide. Any polymer consisting of more than about 10 monosaccharide (ie. glycose) residues joined to each other by glycosidic bonds. A homoglycan (or homopolysaccharide) is composed of only one type of monosaccharide residue, whereas a heteroglycan (or heteropolysaccharide) is composed of two or more different kinds of monosaccharide residue. A homoglycan is generally named after the constituent monosaccharide, with the +ose ending replaced by +an. Glucan is any glycan composed solely of glucose residues; hence, cellulose, dextran, dextrin, and starch are all glucans derived from glucose. Distinguish from glycosan, an old term (not recommended) for any anhydrosugar. |
| glycated hemoglobin |
Or glycohemoglobin; Any derivative of hemoglobin that is formed nonenzymically by reaction at the N (Nitrogen) terminus with glucose. In the normal adult human such derivatives constitute a few percent of the total erythrocyte hemoglobin, the most abundant being hemoglobin A1C, which increases severalfold in concentration in diabetes mellitus. Glycation of other proteins such as lens crystallins, collagen and low-density lipoprotein (LDL) has been implicated in the pathogenesis of diabetic complications. The extent of glycation of hemoglobin is a useful measure of the degree to which the diabetes has been controlled. It is sometimes improperly termed glucosylated hemoglobin or glycosylated hemoglobin. |
| glycated proteins |
Proteins which have undergone glycation. These post-translationally modified proteins have been implicated in the etiology of various diseases such as the development of Alzheimer-type pathology. |
glycation
glycosylation
glucosylation
glycated |
Any (nonenzymic) reaction of glucose (or another sugar) with a peptide or protein where the product is not a glycoside but results from condensation of the sugar with free amino groups to form a Schiff base which then undergoes an Amadori rearrangement.
The term also includes glycosylation, any chemical reaction, whether enzyme-catalyzed or not, that links a glycose to a peptide or protein and thereby forms a glycopeptide or glycoprotein.
Glucosylation is simply the special case of glycosylation when the glycose is glucose.
glycated, having undergone glycation. |
glycemia (glucemia)
normoglycemia
hyperglycemia
hypoglycemia |
The presence of glucose in the blood (but sometimes used for the presence of sugars, in general). When the concentration of glucose in the blood is within normal limits the condition of the blood (or of the corresponding individual) is termed normoglycemia. An abnormally elevated concentration, hyperglycemia, may result in thirst, whereas an abnormally depressed concentration, hypoglycemia, may produce sweating, trembling, hunger, irritability, and in extreme cases, confusion or coma. See also diabetes mellitus, glucose tolerance test. |
glycerol
glycerin(e) |
1,2,3-propanetriol, CH2OHCHOHCH2OH; a sweet hygroscopic, viscous liquid, widely distributed in nature as a constituent of many lipids (see acylglycerol). The older name glycerin(e) is now obsolete in technical usage. |
glyco+
glyc+ |
Comb. form, glyc+ (before a vowel)
1) indicating sweet-tasting.
2a) indicating sugar. 2b) (in medicine) indicating glucose. See also gluco+.
3) (in chemical nomenclature) indicting covalently linked to carbohydrate.
4) indicating combination with glycine; eg. glycocyamine. |
| glycose |
1) A less frequently used term for any monosaccharide.
2) An obsolete name for glucose. See also glyco+ (def. 2))
3) (Sometimes) an alternative name for corn syrup. |
glycoside
glycosyl
aglycon
glucoside |
Any compound formed by the substitution of a glycosyl (monosaccharide sugar) group into a hydroxyl group, a thiol group, or a selenol group of a compound; the resulting compound may be named, respectively, O-glycoside, S-glycoside (thiolglycoside), or Se-glycoside (selenoglycoside).
An aglycon is the non-sugar glycoside moiety.
Glucoside (formerly), any glycoside in which the sugar moiety is a glucose residue. |
half-life
biological half-life
effective half-life |
1) The time for one half of the atoms of an amount of radionuclide to undergo radioactive decay.
2) A similar measure of the stability (ie. rate of change of state) of an excited atom or molecule, a radical, an unstable elementary particle, etc.
3) The time for one half of the amount of an administered substance to be metabolized or excreted. For clarity, any use of the term half-life must also state which body system is being discussed since the substance may simply move from one system to another.
If the substance is radioactive, the time required for one half of the dose to be eliminated biochemically or physiologically is termed the biological half-life and that required for one half to disappear by radioactive decay as well as by the elimination is the effective half-life.
4) The time for one half of the number of cells in a tissue or organ to be replaced by new cells. |
| haploid |
Describing a cell, an organism or a nucleus of a cell having a single genome or a single set of homologous [chromosomes]; ie. containing half the diploid number. Eg. sperm and egg cells. See gametes and meiosis. |
heat shock protein
hsp |
Abbr. hsp. Any of a group of specific proteins that are synthesized by both prokaryotic and eukaryotic cells, eg. bacteria, yeasts, and Drosophila, after they have been exposed to a temperature that is higher than normal. Other stresses, such as free-radical damage, acute strenuous exercise, and calorie restriction have a similar effect. Many members of the hsp family are not induced but are normally present in all cells. The induction of heat-shock proteins within both human cell cultures and whole bodies has in recent years been the topic of much research. It is part of the larger topic of hormesis and is thought to play a major role in the process by which calorie restriction extends the average and maximum lifespan of many animal species. |
hemat+
hemo+
hema+
hemato+
hem+ |
Combining forms, or (before a consonant) hemato+, or (before a vowel) hem+ denoting blood. |
hemopoiesis
hematopoiesis |
The process of blood formation, especially the formation of blood cells, occuring mainly in the bone marrow. |
| helper cell |
Abbr. TH cell. A T-lymphocyte, carrying the surface markers CD4 and CD5 in the human (L3T4 and Ly1 in the mouse), that acts as an inducer of the effector cells for both humoral and cell-mediated immunity. Helper cells recognize and bind to antigen in combination with class II major histocompatibility complex molecules on the surface of antigen-presenting cells, and generate lymphokines that stimulate effector cytotoxic cells and B-cells (formerly  cells). Other cells of the helper phenotype, eg. CD4+ cells, are known as T-suppressor cells and are responsible for suppression, exerting negative feedback control on the helper cells. cells). Other cells of the helper phenotype, eg. CD4+ cells, are known as T-suppressor cells and are responsible for suppression, exerting negative feedback control on the helper cells. |
hepato+
hepat+ |
Combining form, or (before a vowel) hepat+, denoting of or pertaining to the liver. |
| hetero+ |
Combining form 1) other, different, unusual. Compare allo+, homo+ (def. 1), iso+, xeno+.
2) for, from, or directed towards a different species. Compare allo+, homo+ (def. 2), iso+, xeno.
3) containing atoms, groups, linkages, residues, or subunits of different kinds. Compare homo+ (def. 3)
4) being or pertaining to an atom other than the predominant or significant one, especially of a nitrogen, oxygen, or sulfur atom in a ring containing mainly carbon atoms. Compare homo+ (def. 4) |
| histamine |
A compound formed by the decarboxylation of L-histidine and present in many mammalian tissues, with especially high concentrations in lung, skin, and intestine; it is stored in the granules of tissue mast cells and circulating basophil cells. Histamine is a potent vasodilator; it also increases capillary permeability, causes contraction of smooth muscle, plays a role in the regulation of gastric secretion, and acts as a mediator in allergic and anaphylactic conditions |
| histidine |
The trivial name for 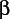 -4-imidazolyalalanine; -4-imidazolyalalanine; 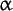 -amino -amino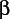 -imidazole-4-propionic acid; a chiral -imidazole-4-propionic acid; a chiral 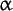 -amino acid. L-Histidine (symbol: H or His) is a coded amino acid found in peptide linkage in proteins. It is also a component of the isopeptide (having an isopeptide bond) carnosine. In mammals it is an essential dietary amino acid and is glucogenic. Histidine is also important as the iron binding site of hemoglobin. D-Histidine (symbol: D-H or DHis), is not known to occur naturally. -amino acid. L-Histidine (symbol: H or His) is a coded amino acid found in peptide linkage in proteins. It is also a component of the isopeptide (having an isopeptide bond) carnosine. In mammals it is an essential dietary amino acid and is glucogenic. Histidine is also important as the iron binding site of hemoglobin. D-Histidine (symbol: D-H or DHis), is not known to occur naturally. |
histo+
hist+ |
Comb. form, or (before a vowel) hist+ denoting tissue. |
| histogram |
A diagram representing a frequency distribution. It consists of a number of contiguous rectangles whose widths are proportional to the class interval under consideration and whose heights are proportional to the frequency associated with each class. (For more, see histogram demo; histogram example (height of a male population).) |
holo+
hol+ |
Comb. form, or (before a vowel) hol+ denoting whole, entire, complete; wholly, entirely, completely. |
homeostasis
homeostatic |
The maintenance of a relatively constant internal environment in the bodies of higher animals by means of a series of interacting physiological and biochemical processes possessing self-regulating negative feedback. -homeostatic, adj. |
| homo+ |
Comb. form 1) the same, similar, alike. Compare allo+, hetero+ (def. 1), iso+, xeno+.
2) from, or directed towards, the same species. Compare allo+, hetero+ (def. 2), iso+, xeno+.
3) containing atoms, groups, linkages, residues, or subunits of the same kind. Compare hetero+ (def. 3).
4) denoting the next higher homologue of a specified chemical compound, especially one whose molecules contain, in a hydrocarbon chain or ring, one more methylene, -CH2-, then those of the parent compound, eg. homocysteine, homoserine, homo-5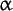 -pregnane. Compare nor+. -pregnane. Compare nor+. |
| homocysteine |
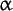 -amino- -amino-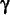 -mercaptobutyric acid; 2-amino-4-mercaptobutanoic acid; an important intermediate in the metabolic reactions of its (S)-methyl derivative, and nutritional metabolic amino acid precursor methionine. Its concentration in human serum is raised after stroke, and high serum concentrations are thought to be risk factors for cardiovascular disease, Alzheimer's disease and many other possible diseases of aging. -mercaptobutyric acid; 2-amino-4-mercaptobutanoic acid; an important intermediate in the metabolic reactions of its (S)-methyl derivative, and nutritional metabolic amino acid precursor methionine. Its concentration in human serum is raised after stroke, and high serum concentrations are thought to be risk factors for cardiovascular disease, Alzheimer's disease and many other possible diseases of aging. |
| homocystine |
2,2'-diamino-4,4'-dithiobis(butanoic acid); the disulfide oxidation product of homocysteine. |
homologous
homology |
1. Having a related or similar position, structure, etc.; corresponding; exhibiting homology (related or similar position or structure).
2. (in chemistry) describing compounds that form a series with successive constant differences in composition. Periodic Table w/ La & Ac series identified
3. (in biology) of common ancestry; especially of organs and tissues that have a similar anatomical position and structure in different species by virtue of their common evolutionary origin, even though their functions may have come to differ; eg. the wing of a bird and the forelimb of a reptile.
4. (in biochemistry) a. (of sequences of residues in encoded macromolecules) having the same or similar residues at corresponding positions.) With respect to proteins, the term is used to imply a common evolutionary origin. Specifically this requires evidence based on gene structure and not merely a similarity of protein structure.
b. (of proteins from different species) having identical or similar functions.
Compare heterologous.
|
homolysis
homolytic |
(In Chemistry) the cleavage of a covalent bond in such a manner that each of the fragments between which the bond is broken retains one of the bonding electrons: eg. A-B -> A• + B•. --homolytic adj.
|
hormesis
hormetic |
An effect where a small quantity of a substance or small application of a process acts in a beneficial manner though a large quantity or application of the same is harmful. Hormetic, pertaining to hormesis. |
| hormone |
Any substance formed in very small amounts in one specialized organ or group of cells and carried (sometimes in the bloodstream) to another organ of group of cells, in the same organism, upon which it has a specific regulatory action. Compare: endocrine. |
| hybrid |
1) Something derived from heterogeneous sources or composed of different or heterogeneous elements.
2) (in genetics) the offspring of two animals or plants of different species, or (less strictly) different varieties.
3) (in chemistry) a bond or valence orbital obtained by the linear combination of two or more different atomic orbitals. |
hydration
hydrate |
1) the act or process by which water molecules surround and bind to molecular entities.
2) the addition of H and OH to a chemical species.
3) the formation of a hydrate, any adduct of water with another compound or element, usually in definite molecular proportions. |
hydro+
hydr+ |
Comb. form, or (sometimes before a vowel) hydr+ 1) indicating or denoting water, liquid, or fluid.
2) indicating the presence of hydrogen in a compound. |
| hydrocarbon |
Any organic compound composed exclusively of hydrogen and carbon; or of, pertaining to, or being a hydrocarbon or a chemical group or side chain composed of hydrogen and carbon only. |
hydrogenate
hydrogenated
hydrogenation |
To undergo or cause to undergo reduction by addition of the hydrogen atoms of dihydrogen, usually with the aid of a catalyst such as finely divided forms of nickel, palladium, or platinum. |
| hydrogen bond |
An association between an electronegative atom, eg. flourine, oxygen, nitrogen, or sulfur, and a hydogen atom attached to another such electronegative atom. |
hydrolysis
hydrolytic
hydrolyze (hydrolyse, Br) |
The rupture (separation into two components) of one or more chemical bonds by reaction with, and involving the addition of, the elements of water. Hydrolytic, of or pertaining to hydrolysis.
Hydrolyze (hydrolyse): to subject to, to undergo, or to effect hydrolysis. |
| hydroxo+ |
Comb. form indicating the presence of a OH- ligand in a coordination compound - |
hydroxyl group
hydroxy+
hydroxylate |
The (covalently linked) -OH group of any compound. Hydroxy used as a prefix, denotes the presence of one or more hydroxyl groups. Hydroxylate, to introduce one or more hydroxyl groups into a compound, often by the replacement of hydrogen atom(s). |
| hygroscopic |
Of a substance tending to absorb water from the surrounding atmosphere. |
| hyper+ |
Prefix indicating over, beyond, excessive, or in excess; above the normal range. Compare hypo+, normo+ |
| hypertonic |
1) (of a solution) having a higher osmotic pressure than that of some given solution, particularly the fluid in or surrounding a given type of cell or a body fluid.
2)(of a muscle) showing or characterized by excessive tone or tension. Compare hypotonic |
| hypo+ |
Prefix, or (before a vowel)hyp+ denoting under, beneath, below, less than, or in deficit; below the normal range. Compare hyper+, normo+ |
| hypotonic |
1) (of a solution) having a lower osmotic pressure than that of some given solution, particularly the fluid in or surrounding a given type of cell or a body fluid.
2)(of a muscle) showing or characterized by diminished tone or tension. Compare hypertonic. |
| hysteresis |
The delay between the effect on a system and the cause producing the effect. |



