Most of the locally defined terms in this glossary are ones that have significant and particular application to human life expansion. Many of them have one or more technical meanings within the sciences related to life expansion that are different from that of the common usage, or even within other sciences. In such cases, we have chosen to include only the meaning(s) relevant to the subject(s) covered within MoreLife. In constructing our definitions we wish to acknowledge the value of these references.
In order to prevent unnecessary duplication, we have selected several excellent sources for external definitions. However, since our usage of some terms is quite specialized and we wish to emphasize certain parts of their meanings, we often locally define terms from these dictionaries within this glossary. In addition, because many anatomical parts and various specific diseases may be of interest to the reader, links to well presented glossary information on other websites have been provided.
| dalton |
Symbol Da; a non-SI unit of atomic and molecular mass, equal to 1/12th of the mass of the nuclide carbon-12. |
| de+ |
Prefix (in organic chemical nomenclature) denoting replacement of a specified group or atom in a specified chemical compound by a hydrogen atom. Exceptions are deoxy+ and dehyrdo+. |
deamination
deaminase
deamidation |
The process of removing an amino group from an organic compound usually by means of a deaminase enzyme catalysing this nonoxidative reaction producing ammonia. Deamidation is the removal of an amino group from an amide. |
decarboxylation
decarboxylase |
The process of removing the carboxyl group from a carboxylic acid as carbon dioxide (CO2) leaving behind the hydrogen atom. This process may sometimes be spontaneous, but any catalytic enzyme involved is called a decarboxylase. |
| deficiency disease |
Any disease resulting from the deficiency of one or more essential nutrients, eg. a vitamin, an essential amino acid, or an essential mineral. |
| dehydration |
1) in physiology, excessive loss of water from the body tissues. Dehydration is accompanied by a disturbance in the balance of essential electrolytes, particularly sodium, potassium, and chloride.
2) in chemistry, the removal of combined, or adsorbed, water from a chemical compound. |
| dehydro+ |
Prefix (in chemical nomenclature) denoting the loss of two hydrogen atoms. In common usage it is often used in place of the more correct, didehydro+; e.g. 7,8-didehydrocholesterol is often termed dehydrocholesterol. |
| denaturation |
(of a protein) A process in which the three dimensional shape of a molecule is changed from its native state without rupture of peptide bonds. (Peptide bond structure) |
| deoxy+ |
Prefix (in organic chemical nomenclature) denoting the replacement of a hydroxyl group by a hydrogen atom. Used especially with hemoglobin, to signify absence or removal of dioxygen. |
| dephosphorylation |
The process of removing one or more phosphoric (ester or anhydride) residues from a molecule. Often accomplished by hydrolysis. |
desaturation
desaturase |
Any process or reaction in which an organic compound becomes unsaturated, eg. by removal of two hydrogen atoms from adjacent carbon atoms.
An enzyme which catalyzes such a reaction is called a desaturase. |
| detoxication |
The act or process of counteracting or rendering innocuous a poisonous substance. |
di+
bis+ |
1) prefix (in chemical nomenclature) indicating the presence in a molecule of two identical unsubstituted groups, eg. diethlsulfide, 1,3-dihydroxyacetone. (distinguish from bis+ (def. 2)) Slightly different from bis+ which simply denotes two, twice, doubled.
2) or bis+ (def. 1) denoting two, twofold, twice, doubled. |
diploid
germ cells
gametes
meiosis |
Describing a cell or nucleus having two sets of homologous chromosomes; ie containing twice the haploid number. In humans, all cells except germ cells (ova and sperm, also called gametes) are diploid, except for the sex chromosomes in males. Meiosis, the process by which the nucleus of a diploid cell divides twice with the formation of four daughter haploid cells, each containing half the number of chromosomes of the parent nucleus. It occurs during the formation of gametes from diploid cells. |
disease
syndrome |
A condition of abnormal vital function involving any structure, part or system of an organism other than one arising directly from physical injury, characterized by a recognizable set of signs and symptoms, and attributable to heredity, infection, diet, or environment. Syndrome, a complex of signs and symptoms resulting from a common cause or appearing, in combination, to present a clinical picture of a disease or inherited abnormality. |
| dismutation |
A chemical reaction in which a single compound serves both as an oxidizing agent and as a reducing agent. |
| disorder |
Although still very commonly used within medical terminology and writings, there is no accepted medical or other technical definition of this term. It is generally used in its standard language meaning of "disturbance", "derangement", "abnormality", etc. although not such that it is strong or clear enough to be called a disease, ie. not characterized by a recognizable set of signs and symptoms. Compare disease. |
D/L convention
D-
L- |
Originally in 1891, a method devised by German chemist Emil Fischer of representing the configuration of each chiral center in glucose in an unambiguous manner; since x-ray confirmation in 1951, a convention of symbols to designate the absolute configuration around a chiral element in 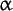 -amino acids, cyclitols, monosaccharides, and derivatives of these classes of compounds. Within these classes it can be applied to any molecule of the type R1CHXR2that can be oriented in a Fischer Projection [use graphic view; slides 6-8] with the most highly oxidized atom at the top; then if X is on the right the configuration is designated by the prefix D- (from the latin dexter meaning right), whereas if X is on the left the configuration is on the left the configuration is designated by the prefix L- (from the latin laevus meaning left). Racemic mixtures of D and L enantiomers are designated by the prefix DL-. -amino acids, cyclitols, monosaccharides, and derivatives of these classes of compounds. Within these classes it can be applied to any molecule of the type R1CHXR2that can be oriented in a Fischer Projection [use graphic view; slides 6-8] with the most highly oxidized atom at the top; then if X is on the right the configuration is designated by the prefix D- (from the latin dexter meaning right), whereas if X is on the left the configuration is on the left the configuration is designated by the prefix L- (from the latin laevus meaning left). Racemic mixtures of D and L enantiomers are designated by the prefix DL-. 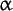 -Amino acids are designated D- or L- according to the configuration at the -Amino acids are designated D- or L- according to the configuration at the 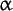 -carbon atom. (See -carbon atom. (See 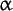 -carbon atom in structural formula of Arginine. -carbon atom in structural formula of Arginine.
More on Fischer Projections (D/L naming convention) [Chime plug-in required for full usage]
To distinguish two enantiomers in an absolute sense, the R-S convention is used. |
down regulation
down-regulated |
1) A decrease in response of a cell to a hormone brought about either by a decrease in receptor number or by the uncoupling of an effector component.
2) A decrease in the activity of an enzyme or the amount of a protein resulting from the action of an effector molecule on gene expression or protein synthesis. |
drug
medicine |
Any substance, other than a nutrient, which is used in the diagnosis or treatment of human disease.
Note: The above is a narrow definition specifically to make the term "drug" well defined and to avoid the philosophical distortion which the term has endured in modern times. However, more generally the term "drug" is defined legally as a medicine, anything applied to the body to treat or prevent a disease or condition. |
| EC50 |
Effective Concentration - 50%. The concentration of an agonist which produces 50% of the maximum possible effect of that agonist. Compare IC50 |
effector,
positive/negative |
Any small molecule or ligand that interacts with an enzyme thereby changing its catalytic behavior but that is not itself changed during the enzyme action. A positive effector enhances catalytic activity while a negative effector reduces it. |
| efficacy |
Although this word is mostly used in its standard language meaning, it also has the following technical definition: a quantitative index of drug or nutrient action related to the magnitude of a tissue response generated at a given level of receptor occupancy, with (full) agonists (of high efficacy) generating the maximum possible response. Drugs (nutrients) with lower efficacy, that cannot generate a maximal response even when all receptors are occupied, are known as partial agonists; antagonists have an efficacy of zero. The overall response to a drug (nutrient) will be a function of its efficacy and receptor binding affinity, which determines the proportion of receptors occupied at a given concentration of drug (nutrient). |
eicosanoid
arachidonic acid
eicosapentaenoic acid |
Any of various agents that are C20 polyunsaturated fatty acids, commonly arachidonic acid (a specific isomer of any straight-chain fatty acid having twenty carbon atoms and four double bonds per molecule), or an eicosapentaenoic acid (any straight-chain fatty acid having twenty carbon atoms and five double bonds per molecule), or their skeletal derivatives. They include the leukotrienes and the prostanoids (prostaglandins, prostacyclins, and thromboxanes) and are noted for their widespread biological activity, such as contraction or relaxation of smooth muscle, platelet aggregation (clumping), and the inflammatory response. |
| elastin |
A major structural protein of mammalian connective tissues, especially elastic fibres, and found at sites such as the aorta, nuchal ligament (at nape of neck), and lung. Glycine makes up one third of the amino-acid residues in elastin; glycine, proline, alanine, and valine together account for over 80% of its residues. The polymeric chains are cross-linked together into an extensible 3-D network. |
| element |
Any basic and distinct component of matter that is not resolvable into simpler components with differing chemical properties. Each consists exclusively of atoms with the same unique proton number. Ninety-three chemical elements are known to occur naturally, either in the free state or in combination with others; to date, a further 16 (?17) have been produced artificially. Periodic Table of Chemical Elements |
emulsion
emulsify |
A temporary or permanent dispersion of an oil or other hydrophobic material in an aqueous solution, or vice versa, forming an oil-in-water emulsion or a water-in-oil emulsion respectively. Emulsify, to form an emulsion. |
endo+ end+ ento+
exo+ |
A combining form meaning "inward, within".
Compare exo - a combining form meaning "outside, outward". |
| endocrine |
Pertaining to a process in which a gland or other group of cells synthesizes hormones and secretes them directly in the blood, lymph or other extracellular fluid. |
| endogenous |
Arising or developing within an organism, tissue, or cell, and excluding any consequences of externally added agents or materials. |
endorphin
endogenous opioid |
Any endogenous peptide with morphine-like activity. The term was originally applied to a general class of substances of morphine-like acclivity postulated to occur in the brain; endogenous opioid is now used to denote any peptide, found in higher organisms, that has morphine-like activity. |
| energy |
The capacity of a system for doing work. Energy may occur in the form of heat, light, movement, sound, or radiation. Human energy is usually expressed as muscle contractions and heat production, made possible by the metabolism of food that originally acquired the energy from sunlight. Chemical energy refers to the energy released as a result of a chemical reaction, as in the metabolism of food. |
| entropy |
A physical quantity and a thermodynamic (relating to supply, use, or acceptance of energy) property indicating the amount of disorder in a system; ie. the amount of energy in a system that is unavailable for doing work. In any irreversible process the total entropy of all systems concerned is increased. In a reversible process the total increase in entropy in all systems concerned is zero, while the increase in entropy, of any individual system, or part of a system, is equal to the heat that it absorbs divided by the thermodynamic temperature. |
| ephedrine |
A structural analogue of epinephrine, it mimics central and peripheral effects of noradrenergic and adrenergic neurons through direct and indirect actions, hence its CNS-stimulant effects and its use as a decongestant in upper respiratory tract infections. |
| epinephrine |
A hormone secreted by the adrenal medulla in response to low blood glucose, exercise, and stress; it is part of the fight-or-flight reaction. It is synthesized by the methylation of norepinephrine. Epinephrine is also a neurotransmitter secreted by neurons in the brainstem. Epinephrine causes a breakdown of glycogen to glucose in the liver, encourages the release of fatty acids from adipose tissue (fat), causes vasodilation of the small arteries within muscle, and increases cardiac output. |
epistemology
epistemological |
Literally: theory of understanding - That branch of philosophy which investigates the nature of knowledge, and the methods by which the mind may gain knowledge of reality including all consistent abstractions from it. Included in epistemology are investigations into the meaning of truth and falsity, and the delimitation of their determinability. epistemological, adj. |
erythro+
erythroblast |
A combining form meaning 'red': erythrocyte, erythroblast (an immature form of a red blood cell, normally found only in bone marrow). |
Escherichia coli
E. Coli |
The sole member of the bacterial genus Escherichia and arguably the most widely used experimental cell system in biochemistry and molecular biology. The organism is present in the intestinal tract of humans and other animals and is common in soil and water. There are numerous strains, some of which are enteropathogenic. |
| essential (nutrient) |
Necessary for adequate or optimal human development and health but either not synthesized by normal human biochemistry or synthesized at an inadequate rate. |
ester
esterfied
esterification |
Any organic compound formed by the elimination of the elements of water between a hydroxyl group of an oxoacid and a hydroxyl group of either an alcohol or a phenol. |
ethyl group
ethane
ethanol
ethyl alcohol
ethene (ethylene) |
The alkyl group, CH3-Ch2-, derived from ethane, CH3-CH3.
Ethanol, formerly called ethyl alcohol, and commonly known as alcohol is a colorless, water miscible, flammable liquid used as a solvent and for human consumption (when produced by fermentation of food sugars). Chemically, ethanol is the combination of an ethyl group with a hydroxyl group, CH3-CH2-OH. Most industrial alcohol is now derived from ethene (ethylene), CH2=CH2, as a by-product in the petroleum industry. |
| etiology |
The cause or causes of disease and their study. |
eugenics
|
The study of methods of improving the hereditary characters of the human race, especially by selective breeding. It is not clear whether eugenics is a rational and worthwhile endeavor even if pursued by entirely voluntary means, but it is certainly philosophically unacceptable if done by force, eg. by government edict. |
| experimental |
Any object, system, or animal group in an experiment which includes, or is subjected to, one of the variables. Compare control. |
express
gene expression
expression vector
expression system
coding
codon
cloned |
(in genetics)
1) To allow or cause the information in a gene to become manifest.
2) To activate the cellular functions involved in gene expression (the process by which the information carried by a gene or genes - the genotype - becomes manifest as the phenotype).
(in cellular DNA technology)
To effect the synthesis of a protein according to the coding (arrangement of DNA and mRNA codons - triplets of nucleic acids) of the corresponding cloned (synthetically replicated or extracted) gene using an expression vector (a vector that promotes transcription of a cloned gene) in an expression system (a cell system into which an expression vector has been introduced and that contains all the enzyme systems needed for translation of messenger RNA). |
| factor |
Any component or cause that contributes to an effect or result; a term often used to denote an uncharacterized (or incompletely characterized) component of a biological system. Compare cofactor. |
| familial |
Describing something transmitted between, or carried by members of a family, especially of a genetic character, condition, predisposition or disease. |
fat
oil |
1) Any triacylglycerol or mixture of triacylglycerols that is solid below 20 °C; those that are liquid at such temperatures are usually referred to as oils.
2) An alternative name for lipid.
3) An alternative name for adipose tissue. |
fatty acid
free fatty acid (FFA)
medium chain triglyceride (MCT) |
Any of the aliphatic monocarboxylic acids that can be liberated by hydrolysis from naturally occuring fats and oils (meaning 1). Fatty acids are predominantly straight-chain (not branched, though they may be bent at various angles depending on the bond structure between carbons) acids of 4 to 24 carbon atoms, which may be saturated or unsaturated. Branched fatty acids and hydroxy fatty acids also occur, and very long chain acids of over 30 carbons can be found in waxes.
FFA is the abbreviation for free fatty acid, a non-esterified (not otherwise joined to another molecule) fatty acid; the term is often used of such acids in the blood.
Free saturated fatty acids with chain length less than 12 carbons are often called Medium Chain Triglycerides (MCTs). |
fatty acid nomenclature
palmitic acid
palmitoleic acid
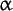 -linolenic acid -linolenic acid
eleostearic acid
linoleic acid
oleic acid
elaidic acid
omega (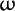 ) carbon ) carbon |
A system of symbols for describing fatty acids. The basic symbolism comprises the number of carbon atoms in the molecule, followed by the number of double bonds; the two numbers are separated by a colon. Thus, 16:0 represents palmitic acid, 16:1 palmitoleic acid, etc. To avoid ambiguity, double bond positions in unsaturated fatty acids should be indicated; 18:3;9,12,15 (also denoted by 18:3 (9,12,15)) represents 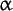 -linolenic acid, 18:3;9,11,13 represents eleostearic acid (a conjugated fatty acid). Double bonds may also be designated by counting from the last carbon in the chain (the omega carbon). Since the Greek letter, -linolenic acid, 18:3;9,11,13 represents eleostearic acid (a conjugated fatty acid). Double bonds may also be designated by counting from the last carbon in the chain (the omega carbon). Since the Greek letter, 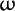 , is often not available for printing, "w" or "n" is often used instead. Thus, , is often not available for printing, "w" or "n" is often used instead. Thus, 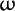 3, w3 and n-3 all describe a fatty acid whose chain has the following structure at the end farthest from the carboxylic acid group: CH3-CH2-CH=CH-, etc. For those common polyunsaturated fatty acids of major nutritional and physiologic significance which have their double bonds successively separated by two single bonds, only the position of the first double bond need be given. Thus, linoleic acid (18:2;9,12) is also represented as 18:2 n-6. 3, w3 and n-3 all describe a fatty acid whose chain has the following structure at the end farthest from the carboxylic acid group: CH3-CH2-CH=CH-, etc. For those common polyunsaturated fatty acids of major nutritional and physiologic significance which have their double bonds successively separated by two single bonds, only the position of the first double bond need be given. Thus, linoleic acid (18:2;9,12) is also represented as 18:2 n-6.
Finally, the orientation of the groups at opposite ends of each double bond may be in either the cis or trans configuration. In the symbolic nomenclature, cis and trans are generally abbreviated to "c" and "t". Thus, for completeness "c" or "t" should precede each group of double bond denoting numbers which have that orientation. For example, oleic acid would be 18:1;c9 whereas elaidic acid would be 18:1;t9. |
fermentation
ferments |
1) The decomposition of chemical substances brought about by ferments - substances or agents that cause fermentation - resulting in the production of simpler substances and often, of energy; an example of this is the anaerobic breakdown of glucose to lactate or ethanol.
2) In biotechnology, the use of microorganisms or cultured cells to produce useful materials, such as antibiotics, beverages, enzymes, and some commodity chemicals. |
ferritin
apoferritin |
Any of a group of electron-dense, major iron-storage proteins that are widely distributed in animal and plant tissues; the iron is stored in a soluble, readily available form. Ferritins consist of a hollow protein shell; the functional molecule, called apoferritin, comprises 24 chains and is roughly spherical, with an external diameter of 12.4-13 nm and an internal diameter of 7-8 nm.
In clinical chemistry, a lowered plasma ferritin is an indication that the body's iron stores have decreased; a concentration < 12µg/L indicates a complete absence of stored iron. |
ferro+
ferri+ |
ferro+ - a combining form denoting containing ferrous (divalent Fe2+ or Fe(II)) iron.
ferri+ - a combining form denoting containing ferric (trivalent Fe3+ or Fe(III)) iron. |
| fiber, dietary |
Plant materials that are resistant to small intestinal digestion. Fiber is composed of the plant cell wall nonstarch polysaccharides. Dietary fiber components consist of two major classes which have different nutritional effects: the water-insoluble celluloses, hemicelluloses, and lignins; and the water-soluble pectic substances, gums (botany), and mucilages. Diseases and disorders attributed to a lack of dietary fiber include: such colonic abnormalities as constipation, diverticular disease, colon cancer, and such systemic abnormalities as hyperlipidemia, cardiovascular disease, diabetes, and obesity. |
fibroblast
fibrocyte |
A flattened, irregular, branched, motile cell found distributed throughout vertebrate connective tissue; also known as a fibrocyte. Such cells form, secrete, and maintain the extracellular collagen and mucopolysaccharide of this tissue. Similar cells occur in many invertebrates. |
fibronectin
opsonization
opsonin |
Any of a group of related adhesive glycoproteins of high molecular mass found on the surface of animal cells, in connective tissue matrices, and in extracellular fluids. They bind to cell surfaces, collagen, fibrin, heparin, DNA, actin, etc. and are involved in cell adhesion, cell motility, opsonization (rendering of microorganisms or other particulate material more susceptible to phagocytosis by coating them with opsonin - a blood serum protein), wound healing, and maintenance of cell shape.
Cellular fibronectin (which is the major cell surface protein of cultured fibroblasts) is greatly reduced or even lost when a variety of cell types are transformed by oncogenic viruses, chemical carcinogens, or temperature-sensitive viruses at the permissive temperature. |
| first messenger |
An alternative term sometimes used for hormone to distinguish it from second messenger. |
fish oil
fish liver oil |
Normally meaning oil obtained from the flesh of fish; it is comparatively rich in fatty acids of the 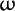 3 family (see fatty acid nomenclature). Generally, the flesh of so-called oily fish, such as herring, mackerel, salmon, and trout which live in colder water, has a higher content of C20 and C22 3 family (see fatty acid nomenclature). Generally, the flesh of so-called oily fish, such as herring, mackerel, salmon, and trout which live in colder water, has a higher content of C20 and C22 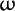 3 acids than that of white fish such as cod or plaice. 3 acids than that of white fish such as cod or plaice.
Oil obtained from the livers of fish also contain these 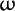 3 fatty acids, but is usually too high in vitamin A (retinol) content to allow enough to be taken to adequately supply them for nutritional purposes. Instead, fish liver oils, particularly from cod and halibut, are used as a nutritional source of vitamin D and vitamin A. 3 fatty acids, but is usually too high in vitamin A (retinol) content to allow enough to be taken to adequately supply them for nutritional purposes. Instead, fish liver oils, particularly from cod and halibut, are used as a nutritional source of vitamin D and vitamin A. |
flavan
flavanone
flavanol
flavone
flavonol |
2-phenylchroman; the parent ring structure on which the structures of flavanols, flavanones, flavones, flavonols, and other classes of aglycons of flavonoids are based.
With respect to the structure of flavan (containing fused ring) shown at the right:
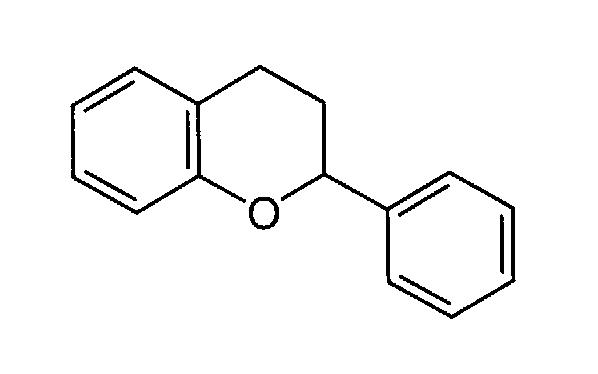
flavanones contain an oxygen atom double bonded to the carbon opposite the ring oxygen.
flavanols contain hydroxyl groups attached to the ring carbons.
flavones have the double bonded oxygen of flavanones and also a double bond between the two carbons on the center ring opposite the double ring connection.
flavonols are flavones which have a hydroxyl group attached to the double bonded inner ring carbon.
These terms are often applied to any hydroxylated derivatives of these basic compounds. |
| flavonoid |
Any of a very large group of water-soluble, phenol derivatives, often brightly colored and usually found in the vacuoles of plant cells. They are glycosides with the structure of their aglycon moieties based on the flavan skeleton, and are classified according to the oxidation state of its pyran ring. 10-12 classes are recognized, but all are based on the parent ring structure of flavan as the aglycon moiety. |
| flux |
The rate of (chemical or physical) transformation of a substance within a living cell or tissue; the rate of translocation of a substance within a living cell or tissue, especially across a cell membrane. Such processes are usually measured in moles (gram-molecules) (symbol, mol) per second. (More on moles (mols)) |
| free |
(in chemistry) Not chemically combined (with another substance); not physically bound (to another substance). |
fructose
fruit sugar
levulose |
symbol: Fru; the trivial name for the ketohexose arabino-2-hexulose; there are two enantiomers, D- and L-fructose. D-(-)-fructose (symbol: D-Fru), commonly known as fructose, and formerly known as fruit sugar, is levorotatory, hence also formerly (and sometimes still) known as levulose; it is the sweetest of the sugars. |
| fused ring |
(in chemistry) any ring of atoms in a molecule that is joined to another ring of atoms in such a way that two atoms are common to both rings. |

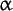 -linolenic acid
-linolenic acid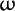 ) carbon
) carbon


